Science That Glows at LSU: Happy 4th of July!
What's better than fireworks? Science that glows! Here in the College of Science we are celebrating independence day not only with pyrotechnics but with fluorescent algae, glowing fish, glow-in-the-dark chemistry and more!
Check out these 7 vibrant examples of science that glows at LSU, one for every day this week! Happy 4th of July!
1. Glow Fish!
Photo of ponyfish and plates of bioluminescent Vibrio bacteria in agar plate. Credit: John Sparks, American Museum of Natural History.
Prosanta Chakrabarty is associate professor and curator of fishes in the LSU Museum of Natural Science. He has studied and described many strange and unique fish species, but one is a group of fishes called ponyfishes, in the family Leiognathidae. These silvery shallow water fish light up, thanks to a unique internal light organ system, producing glowing colors in various parts of their body!
An image Prosanta's former postdoc Matt Davis made while studying in his lab to show the locations of luminesence in ponyfishes.
Ponyfishes have a special light organ containing the Vibrio bacteria Photobacterium leiognathi. These bacteria create light through bioluminescence, a form of chemiluminescence as you might have seen in fireflies, where light is produced by chemical reactions. Ponyfishes use this luminescence to avoid predators and, in some species, to communicate during sexual interactions!
"I traveled across the Indo-West-Pacific studying ponyfishes for many years," Prosanta said. "I first started sequencing the Vibrio bacteria while a student at the University of Michigan, in Paul Dunlap's lab, and then as a postdoc working with John Sparks."
Prosanta has described several new species of ponyfish, including Nuchequula mannusella, through fieldwork in the Philippines and other locations.
Photo of Nuchequlla mannusella found in Taiwan. Credit: Prosanta Chakrabarty.
"We discovered these fish through good old fashioned sleuthing," Prosanta said. "They all look pretty much the same (silvery shiny little fish), but when you examine them more closely, males of many species have translucent patches where light is emitted. These distinctive patches of light attract females. The light can come out the top of the head, the flanks (in a variety of shapes) and through the mouth. The bacteria produce light for reasons that are not well understood. It is probably a by-product of some other function like metabolism."
Read more about the luminescence of ponyfishes here.
2. Cancer-Targeting Compounds that Light Up
Fluorescent compounds in cells. HEp2 cells stained with ER-Tracker (green) and the target compound, CooBn-BODIPY (red). Image credit: Zehua Zhou, LSU.
Charles H. Barré Distinguished Professor of Chemistry Graça Vicente and researchers in her lab including research associate Zehua Zhou create new organic materials that absorb and emit energy, or light, in the visible and near-infrared region of the optical spectrum (400–900 nanometer wavelengths). The compounds are also often fluorescent, emitting longer wavelengths of light after they have absorbed light, allowing researchers to visually confirm delivery of these compounds to the inside of cancer cells grown in the lab. Vicente's lab develops these compounds to serve as biomedical imaging agents, ion sensors and even cancer therapeutics! The compounds absorb energy that can be used to kill cancer cells on the spot.
Zehua works with these fluorescent compounds to ensure that they can enter cancer cells to deliver their therapeutic effects. The various fluorescent compounds produced in the Vicente lab may travel to different cell organelles, or different areas in the cell. Zehua can determine where a compound will tend to locate inside of a cancer cell by imaging cells treated with these compounds via confocal microscopy.
BAM!
3. Glow-in-the-dark Chemistry
Chemistry major Corey Weber is conducting undergraduate research with LSU Professor of Chemistry John Pojman. Corey is studying the effect of sheets of copper on propagating polymerization fronts in QuickCure Clay, a product developed by Dr. Pojman. Polymerization is a chemical reaction where single molecules come together to form polymer chains or three-dimensional networks.
The reaction in QuickCure Clay is started by heating one end of the sculpting clay material. An exothermic (aka heat-releasing) reaction propagates from the source of the heat, hardening the material. In the video above, a glow-in-the-dark powder glows brightest at the location of the exothermic polymerization front and allows visualization of the complex patterns of propagation through the clay.
QuickCure Clay can be used to create sculptures that are otherwise difficult to create, such as lizards standing on their tails!
Photo of a sculpture created by LSU alum and artist Shelby Prindaville, with QuickCure Clay! Credit: Shelby Prindaville.
Speaking of lizards...
4. Lizards with... Green Blood!
A male Prasinohaema prehensicauda in a tree in the southern highlands of New Guinea. Biliverdin causes bright green coloration of the muscles, bones, mucosal lining, and tongue. Credit: Chris Austin, LSU.
Most vertebrates have red blood, due to the iron-rich protein hemoglobin found in their bloodstream. But several species of lizards from the mega-diverse island of New Guinea have evolved bright green blood! Zach Rodriguez, a PhD student studying evolutionary biology in the LSU Museum of Natural Science with Curator of Amphibians and Reptiles Christopher Austin, is studying these green-blooded lizards, including the skink Prasinohaema prehensicauda.
"An unusually high concentration of the green bile pigment biliverdin in the circulatory system of these lizards makes the blood, muscles, bones, tongue, and mucosal tissues bright green in color, eclipsing the crimson color from their red blood cells," Zach said. "However, green blood is a risky trait to have. Bile pigments are toxic waste products of red blood cell recycling, and in all other vertebrates, this causes jaundice often seen in newborns and people with malfunctioning livers. Normally, bile pigments like biliverdin are rapidly removed from the body and are generally undetectable in the blood. However, these lizards keep it at extremely high concentrations in their bodies and have somehow evolved a resistance to biliverdin toxicity."
Biliverdin can have fluorescent properties when bound to other proteins, such that some other organisms with high levels of biliverdin including walleyes (Sander vitreus, a freshwater fish) could glow under UV-light!
Learn more about green-blooded lizards with Zach here.
In this image, one tube (top) shows a blood sample from a regular red-blooded lizard from New Guinea (Lamprolepis smaragdina). The other tube (bottom) reveals the bright green blood of Prasinohaema. The bile in this blood could kill other species, but Prasinohaema handles it just fine. Credit: Chris Austin, LSU.
5. Studying Photosynthesis with Fluorescent Algae
David Vinyard working in his photosynthesis lab - where working in the dark is doing good science! Credit: David Vinyard.
David Vinyard is an assistant professor of biological sciences at LSU. Trained as a chemist, David now studies the molecular mechanisms of photosynthesis, or how plants and some microbes turn light into energy. Through his research, he hopes to reveal nature’s molecular blueprint for sustainable solar-to-chemical energy transduction, and to regulate and improve the efficiency of photosynthesis. In the lab, David is usually found working with cold samples in dark rooms with big magnets.
Image of fluorescent chlorophyll. Credit: David Vinyard.
David Vinyard: "One way to measure photosynthetic efficiency is to quantify fluorescence from chlorophyll. In this set of photographs, two samples of green algae are illuminated by a violet light source. When we turn off the room lights, we see that the sample on the right is glowing bright red. This is the result of chlorophyll molecules absorbing the violet light and fluorescing red light. High chlorophyll fluorescence indicates that less solar-to-chemical energy conversion is occurring and that photosynthetic efficiency is low."
6. Color-Changing Chemistry
Image displaying platinum compounds that change color to detect pollutants. Credit: Siddieg Elsiddieg, LSU.
Siddieg Elsiddieg is a PhD student in the LSU Department of Chemistry working with Philip & Foymae West Distinguished Professor Andrew Maverick. Siddieg has been developing platinum sensing compounds that change color to detect pollutants and environmentally relevant anions such as perrhenate (ReO4-), perchlorate (ClO4-) and nitrate (NO3-).
The compounds all contain platinum, and each one has a positive charge. “Opposites attract,” so when the positively charged platinum compounds bump into something that has a negative charge (called an anion), the compounds produce vibrant colors. "We get different colors with different anions," Andrew Maverick said. "Some of the anions, like nitrate and perchlorate, are environmental hazards, so this work could lead to a simple way to test for anions in environmental samples."
Platinum compounds changing colors in presence of solvents. Credit: Siddieg Elsiddieg, LSU.
The intense colors produced by these platinum compounds in the presence of pollutants occur because of the way the anions help the platinum compounds assemble into long chains. "Charcoal and diamond are both made of carbon, but they look very different because of the way the carbon atoms are put together," Andrew said. "Similarly, platinum compounds one at a time look very different from a chain of platinums stuck together. If chains made from nitrate are a little different from chains made with perchlorate, then you can get two different colors, and that way you can tell them apart."
Other research groups have made compounds like this before, but Siddieg and Andrew's compounds are unusual in all of the ways that they change color. The only drawback is that platinum is expensive, so these compounds are also. "Siddieg has also attempted to make compounds with cheaper metals, but they don’t show color changes anything like the platinum compounds," Andrew said. "Fortunately, you don’t need much platinum to get a color change – just a few milligrams is enough."
7. Dyed Fish Noses!
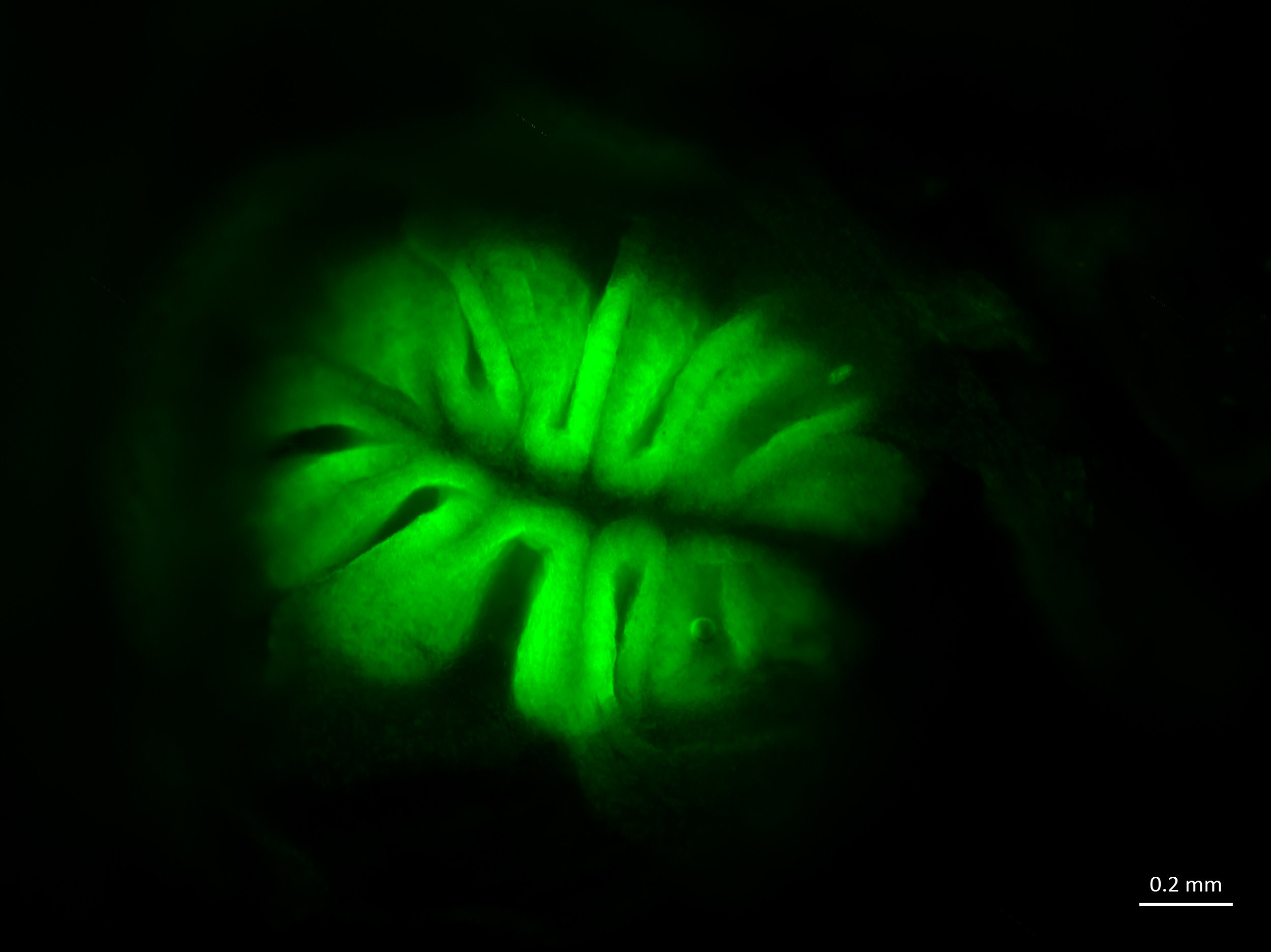
A fluorescent image showing lateral line cells or sensory neuromasts (green dots) in a Mexican blind cavefish. The fish doesn't have eyes, but it can sense water movement with its lateral line system. Credit: Julie Butler, Maruska Lab at LSU.
LSU graduate student Julie Butler studies fish neuroscience with assistant professor Karen Maruska in our Department of Biological Sciences. Yep, they study fish brains! They investigate how fish like the African cichlid fish Astatotilapia burtoni process a range of signals from their environment and other fish, including feel, sound, taste and smell.
Illustration of DASPEI stained ciclid fish. Credit: Julie Butler.
Maruska lab researchers often use a green dye called DASPEI to visualize cells in the brain and nervous system of the fish they study. The fluorescent dye DASPEI can be used to label sensory neuromasts (green dots in the fish image above) to characterize the mechanosensory lateral line system that fish use to detect water movements.
"This dye is taken up only by metabolically active cells," Julie said. "When fish are immersed in it, it primarily gets taken into sensory cells, making them appear bright green under a fluorescent microscope. We use it to visualize the mechanosensory (lateral line) and chemosensory (smell/taste) systems. The lateral line allows fish to detect water movements around them. Mexican blind cavefish don't have eyes - but they have a well-developed lateral line system to help them navigate in complete darkness!"
Maruska lab researchers are studying which regions of the brain are involved in processing lateral line information during social interactions between individual fish, for the first time in any fish species.
Fluorescent image of the olfactory epithelium of a goldfish. Credit: Julie Butler, Maruska Lab at LSU.
Maruska's lab has also used DASPEI to correct misunderstandings about how chemicals affect fish behavior.
"To look at the role of the lateral line system during behaviors such as prey acquisition, predator avoidance and social behaviors, many people use chemicals to kill or disable the cells that detect water movements," Julie said. "Unfortunately, one of the chemicals commonly used to disable to the lateral line, cobalt chloride, also affects chemosensory cells. So these fish can't feel water movements, or smell or taste. To show this, we used DASPEI staining to visualize the olfactory epithelium in several fish species (cichlids, goldfish, and cavefish) and quantified fluorescence intensity (how bright it glows). If the cells are dead, they don't 'glow.' By comparing cobalt treated and control fish, we showed that cobalt treatment also disables cells in the olfactory epithelium. For more than 50 years people have attributed behavioral differences observed after cobalt treatment to the lateral line. But now we know that it may also be taste/smell."
Learn more about LSU Science research by subscribing to this blog below! Happy Independence Day!